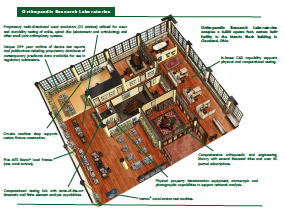
The evolvement of cervical and lumbar disc replacement designs as alternatives to spinal fusion has resulted in a significant number of ongoing United States Food and Drug Administration (USFDA)-sponsored clinical trials. While these seek to establish “safety and effectiveness”, they are of limited in vivo duration and benefit from long-term benchtop comparison. To assure implant durability, mechanical and biological evaluation of these devices, particularly long-term fatigue behavior, is necessary. Both the ASTM and ISO have provided guidance in the evaluation of the performance of artificial spinal discs. These guides propose the biochemical environment, motions, and loading appropriate to simulate long-term use of prostheses employed in total disc arthroplasty. The parameters evaluated include wear measured by gravimetric weight loss, as well as, changes in the articular surface shape and roughness to the extent that these may influence function. This handout describes the wear characteristics of an articulating cervical disc replacement during approximately 80 years of simulated loading in an electro-mechanical multi-axial spinal disc simulator with comparison to clinical retrievals. These results are useful in demonstrating the safety and effectiveness of this device and also present a pre-clinical evaluation methodology for future disc replacement designs.
The evolvement of cervical and lumbar disc replacement designs as alternatives to spinal fusion has resulted in a significant number of ongoing United States Food and Drug Administration (USFDA)-sponsored clinical trials. While these seek to establish “safety and effectiveness”, they are of limited in vivo duration and benefit from long-term benchtop comparison. To assure implant durability, mechanical and biological evaluation of these devices, particularly long-term fatigue behavior, is necessary. Both the ASTM and ISO have provided guidance in the evaluation of the performance of artificial spinal discs. These guides propose the biochemical environment, motions, and loading appropriate to simulate long-term use of prostheses employed in total disc arthroplasty. The parameters evaluated include wear measured by gravimetric weight loss, as well as, changes in the articular surface shape and roughness to the extent that these may influence function. This handout describes the wear characteristics of an articulating cervical disc replacement during approximately 80 years of simulated loading in an electro-mechanical multi-axial spinal disc simulator with comparison to clinical retrievals. These results are useful in demonstrating the safety and effectiveness of this device and also present a pre-clinical evaluation methodology for future disc replacement designs.
Clinical Retrieval and Simulator Comparison of an Investigational Cervical Disc Replacement: An A Priori Requirement
Mar 8, 2010 | Spine